Nuclear Medicine
Nuclear Medicine is a medical specialty that uses targeted radiopharmaceuticals to image and treat diseases. Targeted radiopharmaceuticals contain radioactive particles called radioisotopes.
Quick links: Nuclear Medicine Diagnostic Imaging, Nuclear Medicine Therapy
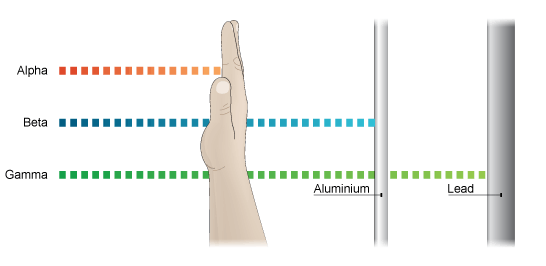
The process of attaching a radioisotope to a molecule is known as labeling or radiolabeling. Different radioisotopes can emit different types of radiation such as alpha, beta, or gamma, each of which has different properties and is used in different clinical settings. Alpha and beta radiation don’t travel very far but are powerful. They are used primarily for treatment as they can damage and kill diseased cells. Gamma radiation travels farther and can be detected using specialized cameras.
Source: http://www.bbc.co.uk/…
Alpha Particles
Alpha particles have a very limited ability to penetrate other materials. These particles can be blocked by a sheet of paper, skin, or even a few inches of air.
Beta Particles
Beta emitters are used in medical applications. Beta particles are lighter than alpha particles, travel a few feet in the air, and can penetrate skin. A thin sheet of metal or plastic or a block of wood can stop beta particles.
Gamma Rays
Gamma rays consist of high-energy waves that can travel great distances at the speed of light and generally have a great ability to penetrate other materials. For that reason, gamma rays are often used in medical applications.
Source: https://www.nrc.gov/…
Targeted nuclear medicine can be divided into two basic categories: imaging and therapy.
Nuclear Medicine Diagnostic Imaging
Nuclear Medicine Imaging often enable physicians to accurately diagnose and stage complex diseases – including cancer, and cardiovascular and neurological disorders in their early stages – as well as monitor disease progression or response to treatment.
Also referred to as molecular imaging or functional imaging, these diagnostic procedures provide detailed pictures of what is happening inside the body at the molecular and cellular level. Where other diagnostic imaging procedures (such as x-rays and computed tomography (CT)) offer pictures of physical structure, nuclear medicine imaging allows physicians to see how the body is functioning and to evaluate its chemical and biological processes.
In nuclear medicine imaging procedures, patients are injected with a radiopharmaceutical agent and imaged with PET (Positron Emission Tomography) or SPECT (Single Photon Emission Computed Tomography) cameras that read the emissions given off by the radiopharmaceutical agent.
PET is an imaging technique that involves detection of a pair of gamma rays emitted from a patient’s body following administration of a radiopharmaceutical that binds to target cells. Fluorine 18 (F-18) and Gallium 68 (Ga-68) are commonly used radioisotopes in PET imaging.
SPECT is another molecular imaging technique. SPECT imaging involves detection of single gamma ray emissions from a patient’s body following administration of a radiopharmaceutical that often binds to a target cell. Technetium 99 (Tc-99) is a commonly used radioisotope in SPECT imaging.
Both PET and SPECT techniques are used broadly in nuclear medicine today. Various factors may determine which imaging modality is used for a specific patient or disease, including availability of a PET or SPECT camera and/or the associated radiopharmaceuticals required to perform the procedures, as well as individual physician or facility practice and preference.
At Advanced Accelerator Applications, we specialize in the development of precision imaging radioligands for oncology, which target specific tumor markers.
Nuclear Medicine Therapy
Targeted Radioligand Therapy combines a precision targeting compound with a therapeutic radioisotope (a radioactive particle). After administration into the bloodstream, the radioligand binds to the tumor and acts as the delivery agent for treatment via the radioisotope, which damages tumor cells, disrupting their ability to replicate and/or triggering cell death.
Many different types of targeting ligands can be used for RLT. One example would be a peptide, which might also be referred to as Peptide Receptor Radioisotope Therapy (PRRT).
The radioisotopes used in nuclear medicine imaging and therapy typically have very short periods of activity, or half-lives, often hours or a couple of days. This means that each dose is produced on-demand for an individual patient. It also means that shipping, handling and administering these medicines requires special procedures. Advanced Accelerator Applications manages the entire process from clinical development and regulatory approval to full commercial production and the logistical management of the delivery process in time for treatment to be effectively administered.